Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Hirade, Tetsuya; Lee, J.; Oka, Toshitaka
no journal, ,
The longest positron annihilation lifetimes, i.e. ortho-positronium lifetimes, in almost all liquids show larger lifetime at higher temperatures. However, only water shows opposite temperature dependence. It is caused by larger possibility of reactions between ortho-positronium and radical species formed in the terminal spur, i.e. the end of the positron track. We have successfully obtained the temperature dependence of the reactions by use of the positron annihilation age-momentum correlation measurement that is the best method to observe ortho-positronium reactions.
Maekawa, Masaki; Kawasuso, Atsuo; Fukaya, Yuki; Yabuuchi, Atsushi
no journal, ,
no abstracts in English
Kuramochi, Akihiko
no journal, ,
no abstracts in English
Yoshida, Tadayoshi; Tsujimura, Norio
no journal, ,
no abstracts in English
Tsujimura, Norio; Yoshida, Tadayoshi
no journal, ,
no abstracts in English
Tago, Takanori; Nagasawa, Naotsugu; Tamada, Masao; Kudo, Hisaaki*; Katsumura, Yosuke*
no journal, ,
no abstracts in English
Tsukui, Narutaka; Watanabe, Shigeki; Yamada, Keiichi*; Hanaoka, Hirofumi*; Oku, Hiroyuki*; Matsuo, Ichiro*; Endo, Keigo*; Ishioka, Noriko
no journal, ,
no abstracts in English
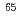 Zn
ZnSuzui, Nobuo; Yamazaki, Haruaki; Kawachi, Naoki; Ishii, Satomi; Ito, Sayuri; Ishioka, Noriko; Fujimaki, Shu
no journal, ,
no abstracts in English
Nakamura, Shinichi*; Suzui, Nobuo; Ito, Sayuri; Kawachi, Naoki; Rai, Hiroki*; Hattori, Hiroyuki*; Chino, Mitsuo*; Ishioka, Noriko; Fujimaki, Shu
no journal, ,
no abstracts in English
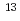 N-labeled nitrogen gas
N-labeled nitrogen gasIshii, Satomi; Suzui, Nobuo; Ito, Sayuri; Ishioka, Noriko; Kawachi, Naoki; Otake, Norikuni*; Oyama, Takuji*; Fujimaki, Shu
no journal, ,
no abstracts in English
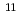 C-labeled photoassimilate
C-labeled photoassimilateYamazaki, Haruaki; Suzui, Nobuo; Kawachi, Naoki; Ishii, Satomi; Ito, Sayuri; Shimada, Hiroaki*; Ishioka, Noriko; Fujimaki, Shu
no journal, ,
no abstracts in English
Kawasuso, Atsuo; Yabuuchi, Atsushi; Maekawa, Masaki; Hasegawa, Shigehiko*; Zhou, Y.-K.*; Asahi, Hajime*
no journal, ,
no abstracts in English
Fukaya, Yuki; Kawasuso, Atsuo; Ichimiya, Ayahiko*
no journal, ,
no abstracts in English
Yabuuchi, Atsushi; Maekawa, Masaki; Kawasuso, Atsuo
no journal, ,
Recently, the SCC propagation model that the vacancy-type defects that accumulate in the crack tip play a role as crack nucleus was proposed. However, there is no information available on the generation or accumulation mechanism of such vacancies around the SCC crack tips. In this study, lattice defect spatial distributions around the SCC crack tip in an austenitic stainless steel have been evaluated probed by positron microbeam. As a result, the increase of S-parameter was observed around the SCC crack. To investigate what type of defect has caused the increase of this S parameter, the  -ray energy distribution spectrum obtained from the surrounding of the SCC crack was compared with the spectrum obtained from the tensile test specimen. From the comparison of these two spectra, it can be concluded that the increase of S-parameter near the SCC crack was caused by plastic deformation induced vacancies.
-ray energy distribution spectrum obtained from the surrounding of the SCC crack was compared with the spectrum obtained from the tensile test specimen. From the comparison of these two spectra, it can be concluded that the increase of S-parameter near the SCC crack was caused by plastic deformation induced vacancies.
 W-
W- Re generator using PZC
Re generator using PZCKurihara, Yuichi*; Nogawa, Norio*; Hashimoto, Kazuyuki; Koike, Yuya*; Morikawa, Naotake*; Ijiri, Kenichi*
no journal, ,
no abstracts in English
Peng, J.; Hao, Y.*; Hu, S.*; Lin, M.; Katsumura, Yosuke
no journal, ,
The thermal stability of 1-allyl-3-methylimidazolium chloride ionic liquid ([Amim]Cl) was studied by using isothermal thermogravimetric analysis and TG/MS. In addition, the radiation stability of [Amim]Cl was investigated by using UV and NMR. During the heating process, the allyl side chain of [Amim]Cl appears to associate with anion Cl and dissociate from imidazole ring with equal probability as the methyl group does. During the  irradiation, although the darkening of color in irradiated [Amim]Cl was observed, the chemical structure of [Amim]Cl is stable at dose of 500 kGy under nitrogen atmosphere.
irradiation, although the darkening of color in irradiated [Amim]Cl was observed, the chemical structure of [Amim]Cl is stable at dose of 500 kGy under nitrogen atmosphere.
Wan, L. K.*; Peng, J.; Lin, M.; Muroya, Yusa*; Katsumura, Yosuke
no journal, ,
The transient absorption spectra of crown ethers in aqueous solution are studied using KrF (248 nm) laser photolysis. Moreover, the rate constants for the reaction of SO
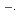 radicals with crown ethers are determined. By laser photolysis, the rate constants of the reactions of SO
radicals with crown ethers are determined. By laser photolysis, the rate constants of the reactions of SO
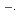 radical with 18-crown-6(18C6), 15-crown-5(15C5) and 1,4-dioxane, have been determined to be 2.5
radical with 18-crown-6(18C6), 15-crown-5(15C5) and 1,4-dioxane, have been determined to be 2.5 10
10 , 2.2
, 2.2 10
10 and 4.2
and 4.2 10
10 M
M s
s , respectively. The values increase with increasing the size of the ethers and K
, respectively. The values increase with increasing the size of the ethers and K and Na
and Na has no obvious effect on the reactivity.
has no obvious effect on the reactivity.
Yamashita, Shinichi; Maeyama, Takuya*; Baldacchino, G.*; Muroya, Yusa*; Taguchi, Mitsumasa; Kimura, Atsushi; Louit, G.*; Katsumura, Yosuke; Murakami, Takeshi*
no journal, ,
no abstracts in English
Lin, M.; Katsumura, Yosuke*; Muroya, Yusa*; Yamashita, Shinichi; Yan, Y.*; Hata, Kuniki; Peng, J.
no journal, ,
With a re-evaluation of the extinction coefficients of solvated electron in 1,2-ethanediol (12ED), 1,2-propanediol (12PD), 1,3-propanediol (13PD), and glycerol (GLY), the time-dependent radiolytic yields of the solvated electron in these diols from the picosecond to the microsecond time range are described. The radiolytic yield of solvated electron in these viscous solvents is found to be quite different from that in aqueous solution. The temperature dependent absorption spectra of the solvated electron in 12ED, 12PD, 13PD, and GLY have been also investigated. Very interestingly, the red-shift of the spectrum with increasing temperature corresponds very well with the spectral shift of electron solvation obtained by femtosecond laser photolysis.
Yan, Y.*; Katsumura, Yosuke*; Lin, M.; Muroya, Yusa*
no journal, ,
The temperature and solvent structure effects on the absorption of solvated electron in some primary alcohols, diols and triols have been investigated. The absorption maximum (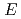
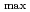 ) shifts to lower energy with increasing the temperature. But at a fixed temperature, the
) shifts to lower energy with increasing the temperature. But at a fixed temperature, the 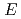
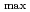 is higher for the solvents having more OH groups. For the primary alcohols having only one OH group, temperature coefficient (-d
is higher for the solvents having more OH groups. For the primary alcohols having only one OH group, temperature coefficient (-d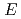
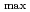 /dT) increases with increasing the chain length. For the same chain length, the temperature coefficient is smaller for the solvents having symmetric molecular structure.
/dT) increases with increasing the chain length. For the same chain length, the temperature coefficient is smaller for the solvents having symmetric molecular structure.